Organizations I've worked with
 NIH
NIH
 USAID
USAID
 Medicaid / CMS
Medicaid / CMS
 U.S. Office of Personnel Management
U.S. Office of Personnel Management
 University of Cambridge
University of Cambridge
 Egg Advisor
Egg Advisor
Health Tech Product & Strategy · Policy · International Markets
I help organizations working on health care access and quality solve their biggest operational blockers — bridging policy, research, and product across the U.S. and Europe. Based in Munich. Work-authorized in Germany and the U.S.
Organizations I've worked with
 NIH
NIH
 USAID
USAID
 Medicaid / CMS
Medicaid / CMS
 U.S. Office of Personnel Management
U.S. Office of Personnel Management
 University of Cambridge
University of Cambridge
 Egg Advisor
Egg Advisor
Across government, research, and health tech — here's what I've actually moved.
Saved tens of thousands in government hours at NIH by overhauling the project management system for communications approvals — cutting turnaround from months to days or weeks across 50+ diverse research sites nationwide.
Co-wrote the pediatric study protocol for NIH's All of Us Research Program — one of the few genomics initiatives to include healthy participants under 18, shaping research design for a million-person biobank.
Managed policy implementation deliverables across all 50 states and territories at Medicaid — building a tracking system for 7 senior-led working groups that eliminated duplicate work and accelerated decision-making.
Rebuilt Egg Advisor's digital presence from scratch — relaunching social media, building a new storefront website, and now overseeing the company's first B2B product launch.
Delivered health and English education to 700+ students in Ecuador as co-founder of EduAmistad, while training 20 health education fellows in evidence-based, inclusive practice.
From defining your product roadmap to building the systems that get it to market — I bring operational rigor and policy fluency to early and scaling health tech teams.
I translate complex U.S. and international health policy into actionable guidance for product, compliance, and leadership teams. I've done this across federal agencies and 50 states.
Fluent in English, Spanish, and Mandarin; studying German. Experienced across the U.S., UK, Ecuador, and Germany. I help organizations build programs and partnerships that work across cultures and health systems.
Egg Advisor — Remote (Fractional Director of Product)
Egg Advisor had no working storefront and couldn't reach its target audience. Without a functional commercial presence, even a strong product had nowhere to land.
Relaunched the company's social media channels, built a new website that now functions as the primary storefront, and took ownership of the full B2B product launch strategy — from positioning to execution planning.
A functioning commercial presence, a live brand channel, and a B2B product launch in progress — built from zero.
National Institutes of Health — All of Us Research Program
NIH's All of Us Research Program was promoting a genomics initiative across 50+ sites with highly varied needs and audiences. Communications approvals were taking months, burning government time and slowing research momentum.
Overhauled the project management system for communications approvals, reducing turnaround from months to days or weeks. Oversaw quality, consistency, and equity across all materials. Also co-wrote the study protocol for the pediatric arm — one of the only genomics programs to include healthy participants under 18.
Tens of thousands of dollars saved in government man-hours. Faster, more consistent communications across a 1M+ participant program. A pediatric protocol that broke new ground in genomics research.
Snapshots from projects I've led.
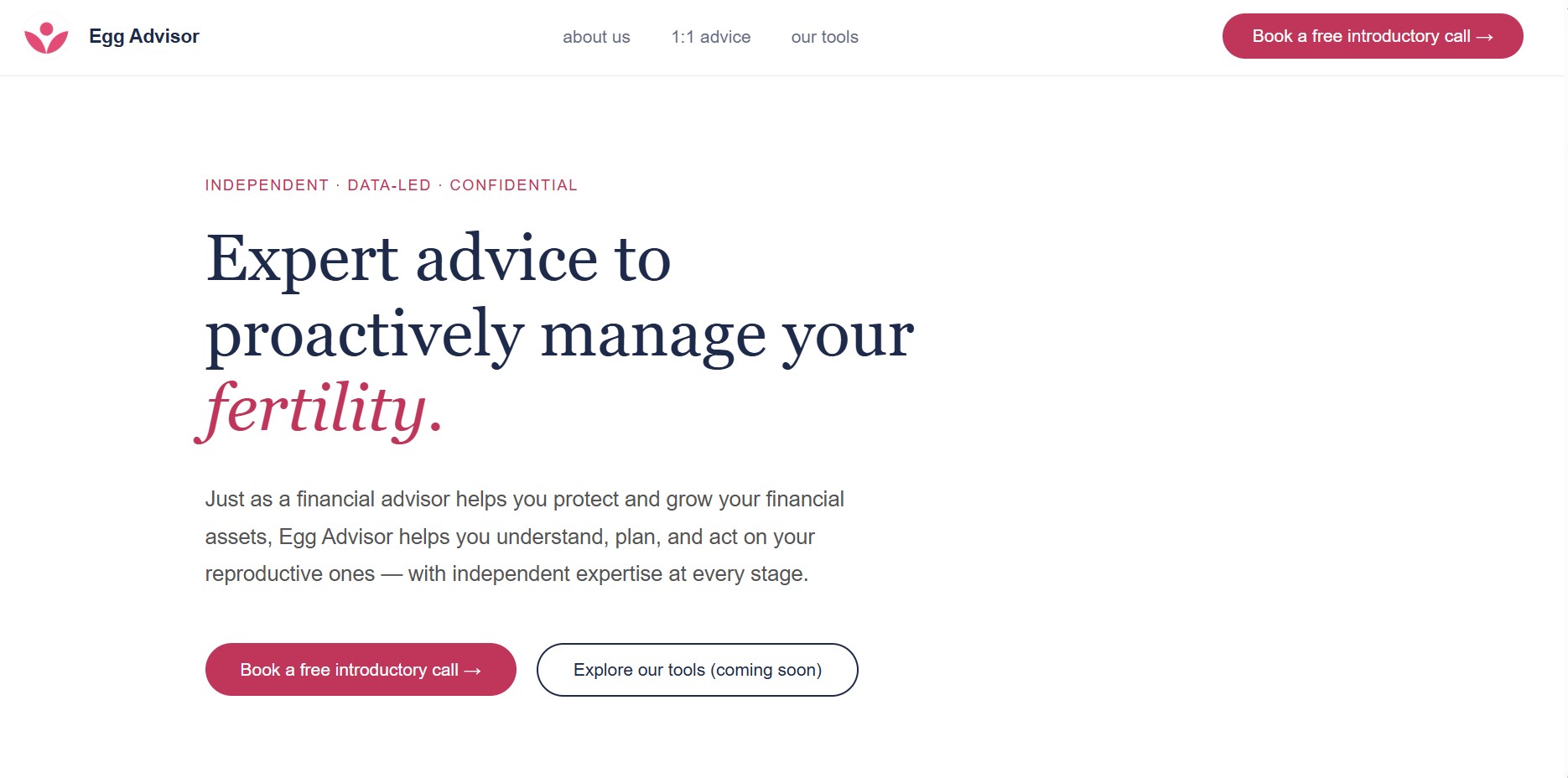
I built a new website with an inviting storefront to re-launch Egg Advisor's services.

I reviewed and approved all materials for a bus that travelled across the U.S. recruiting research participants.

I built a nonprofit that provided health education programming in Guayaquil, Ecuador.
Rebuilt commercial presence from scratch. Overseeing B2B product launch strategy, website, and social media.
Built a cross-functional tracking system for 7 senior-led working groups. Managed policy implementation deliverables at national scale, reducing duplicate work and improving decision-making timelines.
Oversaw communications quality for 50+ diverse research sites. Saved tens of thousands of dollars in government hours by restructuring the communications approvals process. Co-wrote the pediatric study protocol. Managed partnerships with Amazon and CVS.
Managed overseas strategy through analysis of global health, education, and geopolitical data. Led COVID-19 communications between Washington and international missions during the early pandemic.
Designed and delivered federal training programs in Human-Centered Design and systems thinking. Standardized quality and evaluated program effectiveness using mixed methods research.
Co-founded and managed an international nonprofit delivering health and English education to 700+ youth. Trained 20 health education fellows in inclusive, evidence-based practice.
Conducted clinical research across three major New York institutions — supporting studies in nutrition, population health, and Vitamin D deficiency. Facilitated multilingual participant communication in English, Spanish, and Mandarin.
 Master's degree — Education, Globalisation and International Development
Master's degree — Education, Globalisation and International Development
2016–2017 · Cambridge, UK
 B.A. — Chinese Language & Literature, Public Policy, Premed
B.A. — Chinese Language & Literature, Public Policy, Premed
2012–2016 · New York, NY
Macaulay Honors Program — full merit scholarship for exceptional students across the CUNY system
 Published Research — Health and Education Interdependence
Published Research — Health and Education Interdependence
2020
Sabates, R., & Yardeni, A. — Social Determinants of Health and Education: Understanding Intersectionalities During Childhood. pp. 13–33.

My career has moved across research labs, federal agencies, and early-stage startups — but the common thread has always been the same: finding the operational or strategic blockers that stop good health work from scaling, and removing them.
I've managed million-dollar research communications, co-written clinical protocols, advised senior government officials, and launched products from scratch. I bring systems thinking to every level of an organization — and I speak the language of scientists, policymakers, and product teams equally.
Now based in Munich, I'm rooted in the European health landscape while staying connected to the U.S. system I spent a decade inside.
Medicaid, federal implementation, regulatory compliance
Protocol design, IRB compliance, biobanking, multilingual data
Launch planning, B2B, storefront, roadmap development
Equity-centered, multilingual, cross-sector stakeholders
PM overhauls, workflow design, cross-functional coordination
US, UK, China, Germany, Ecuador — 4 languages, multiple health systems
I'm open to roles in health tech product and strategy, health policy advisory, and international health — in Germany, across Europe, or in the U.S. Work-authorized in both. If you're building something that matters, I'd like to hear about it.
Your message goes directly to me. I'll get back to you within 2–3 business days.
Message received!
Thanks for reaching out. I'll be in touch within a couple of business days.